United States Anesthesia Drugs Market Size and Forecast 2025–2033
Rising Surgical Procedures and Advancing Drug Technologies Fuel Market Growth
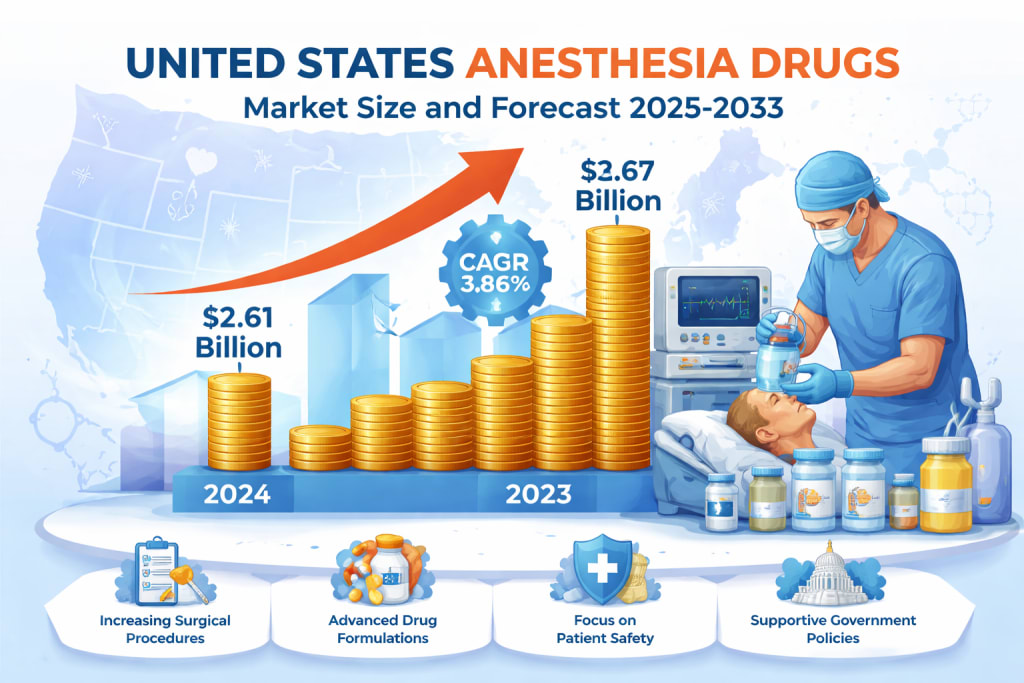
The United States Anesthesia Drugs Market is poised for steady growth over the next decade as healthcare systems continue to expand surgical capabilities and prioritize patient safety. According to Renub Research, the market is projected to increase from US$ 2.61 billion in 2024 to approximately US$ 3.67 billion by 2033, growing at a compound annual growth rate (CAGR) of 3.86% from 2025 to 2033. This growth reflects a combination of rising surgical procedures, technological advancements in anesthetic drug formulations, and increasing emphasis on patient comfort and safety.
Anesthesia drugs are an essential component of modern medicine, enabling surgeons and physicians to perform complex procedures without causing pain or distress to patients. As surgical demand increases across hospitals, outpatient centers, and specialty clinics, the need for reliable and effective anesthesia solutions continues to grow.
Understanding the Role of Anesthesia Drugs in Healthcare
Anesthesia drugs are medications designed to induce anesthesia—a temporary and controlled loss of sensation or consciousness during medical procedures. These drugs allow physicians to perform surgeries and diagnostic interventions safely while minimizing patient discomfort.
There are three main categories of anesthesia:
General anesthesia, which renders the patient unconscious and unaware during surgery
Regional anesthesia, which blocks sensation in a specific area of the body
Local anesthesia, which numbs a small and targeted part of the body
Each type of anesthesia is used depending on the complexity of the procedure and the patient’s medical condition. Commonly used anesthesia drugs include propofol, sevoflurane, lidocaine, and ketamine, each offering different benefits in terms of onset time, duration, and recovery speed.
In the United States healthcare system, anesthesia drugs are widely used across hospitals, ambulatory surgical centers, dental clinics, and specialized medical facilities. They support a broad range of medical applications including surgical operations, childbirth procedures, diagnostic tests, and pain management treatments.
The continued evolution of anesthesia technology has improved drug safety profiles, enhanced patient recovery times, and increased overall clinical efficiency.
Rising Surgical Procedures Driving Market Expansion
One of the primary drivers of the anesthesia drugs market in the United States is the increasing number of surgical procedures performed each year. As medical technology advances and healthcare accessibility improves, more patients are undergoing surgical treatments for a wide variety of conditions.
It is estimated that approximately 15 million surgical procedures are performed annually in the United States, including both elective and emergency operations. These procedures range from orthopedic surgeries and cardiovascular treatments to cosmetic and reconstructive surgeries.
The growing aging population in the United States is another key factor contributing to higher surgical volumes. Older adults often require surgical interventions for conditions such as joint replacement, heart disease, and neurological disorders. As a result, the demand for safe and efficient anesthesia drugs continues to increase.
Minimally invasive surgical techniques are also gaining popularity, as they reduce recovery time and hospital stays. These procedures frequently rely on advanced anesthesia solutions that allow for rapid induction and quicker patient recovery.
According to the American Society of Aesthetic Plastic Surgeons, more than 26.2 million surgical procedures were performed in the United States in 2023, highlighting the significant demand for anesthetic drugs across healthcare settings.
Innovations in Anesthesia Drug Formulations
Advancements in anesthesia drug formulations have played a critical role in shaping the market. Pharmaceutical companies are investing heavily in research and development to create safer and more efficient anesthesia drugs that minimize complications and improve patient outcomes.
Modern anesthesia agents are designed to provide:
Faster onset of action
Shorter recovery times
Reduced side effects
Greater control over anesthesia depth
Drugs such as propofol and sevoflurane have become widely used due to their effectiveness and favorable safety profiles. These medications allow anesthesiologists to precisely control sedation levels during surgery while enabling faster post-operative recovery.
In addition, innovations in local anesthetics have expanded their use in outpatient procedures and pain management therapies. Targeted anesthesia solutions allow physicians to perform minor surgeries and diagnostic procedures without requiring full general anesthesia.
A notable example of product innovation occurred in April 2024, when Baxter introduced Ropivacaine Hydrochloride Injection, USP in the United States. This prefilled, single-dose infusion bag is designed for local and regional anesthesia during surgical procedures and for the management of acute pain in adult patients. The launch strengthened Baxter’s pharmaceutical portfolio while enhancing convenience and safety for healthcare providers.
These types of innovations are expected to drive continued adoption of advanced anesthesia drugs across healthcare institutions.
Growing Focus on Patient Safety and Comfort
Patient safety has become a major priority within the U.S. healthcare system, and anesthesia drugs play a crucial role in achieving safe surgical outcomes. Hospitals and surgical centers increasingly prioritize medications that reduce adverse effects and improve the patient experience.
Common anesthesia-related side effects may include nausea, dizziness, prolonged sedation, and respiratory complications. As a result, healthcare providers seek drugs that provide precise control over sedation levels while minimizing these risks.
New-generation anesthesia drugs are designed to offer:
Improved safety profiles
Faster patient recovery
Reduced post-operative complications
Enhanced comfort during procedures
Patient-centered healthcare initiatives are also encouraging individualized anesthesia approaches tailored to each patient’s medical history and procedure type. This trend is promoting the development of innovative drugs that balance safety, efficiency, and comfort.
With healthcare providers striving to improve surgical outcomes and patient satisfaction, demand for modern anesthetic medications continues to rise.
Government Policies Supporting Market Growth
Government policies and healthcare reforms significantly influence the anesthesia drugs market in the United States. Public healthcare programs such as Medicare and Medicaid provide coverage for millions of surgical procedures each year, creating sustained demand for anesthesia medications.
Federal investments in healthcare infrastructure and surgical technologies have also expanded the number of procedures performed nationwide. As healthcare facilities modernize and surgical capacity increases, the use of anesthesia drugs naturally grows alongside these developments.
Government support also extends to research initiatives focused on improving surgical outcomes and patient safety. These initiatives encourage the development of innovative anesthesia techniques, drug formulations, and delivery systems.
Furthermore, regulatory oversight plays a critical role in ensuring the safety and effectiveness of anesthesia drugs. The U.S. Food and Drug Administration (FDA) maintains strict guidelines for drug approvals, clinical testing, labeling requirements, and dosage recommendations.
These regulations help ensure that only safe and effective anesthetic drugs are introduced to the market, thereby protecting patient health while maintaining high medical standards.
A significant regulatory milestone occurred in March 2023, when Caplin Steriles, in partnership with Lupin Ltd, received FDA approval to market Rocuronium Bromide Injection USP and Thiamine Hydrochloride Injection USP. Rocuronium Bromide Injection is widely used during rapid sequence intubation to facilitate muscle relaxation during surgical procedures.
Following the U.S. launch of Rocuronium Bromide Injection in November 2023, Lupin Ltd experienced a 3% increase in stock value, illustrating how regulatory approvals can significantly influence market dynamics.
Recent Developments in the United States Anesthesia Drugs Market
The anesthesia drugs market continues to evolve with new product launches and regulatory approvals.
In August 2024, Amneal Pharmaceuticals, Inc. received FDA approval for its Propofol Injectable Emulsion USP, which will be available in three concentrations in single-dose vials. Propofol is one of the most widely used anesthetic agents for both induction and maintenance of anesthesia during medical procedures.
Earlier, in February 2024, Hikma Pharmaceuticals PLC launched Fentanyl Citrate Injection, USP in the United States. This short-acting opioid analgesic is commonly used for anesthesia, premedication, induction, maintenance, and post-operative pain management.
These developments demonstrate the continuous innovation occurring within the anesthesia drugs sector, as pharmaceutical companies work to improve drug effectiveness and accessibility.
Market Segmentation Overview
The United States anesthesia drugs market can be segmented based on drug type and formulation.
By Type
The market is divided into three major categories:
General Intravenous Anesthesia
General Inhalation Anesthesia
Local Anesthesia Drugs
General Intravenous Drugs
Key intravenous anesthetic drugs include:
Propofol
Etomidate
Ketamine
Barbiturates
These medications are commonly used for the induction of anesthesia during surgical procedures.
General Inhalation Drugs
Inhalation anesthetics include:
Sevoflurane
Desflurane
Isoflurane
Other inhalation anesthetics
These drugs are typically used to maintain anesthesia during surgery.
Local Anesthesia Drugs
Local anesthetics are widely used in minor surgical procedures and pain management. Major drugs in this category include:
Bupivacaine
Ropivacaine
Lidocaine
Chloroprocaine
Prilocaine
Benzocaine
Other local anesthetics
Reimbursement Landscape in the United States
Reimbursement policies play a key role in shaping the anesthesia drugs market. The market is supported by both private and public health insurance providers.
Private Health Insurance Providers
Major private insurance companies covering anesthesia-related procedures include:
Aetna International
Coventry Health Care
Anthem Blue Cross and Blue Shield
Blue Cross and Blue Shield of Texas
United Healthcare
Public Health Insurance Providers
Government-backed programs supporting anesthesia procedures include:
Medicare
MDWise
These reimbursement systems ensure that patients can access necessary surgical procedures and anesthesia services without excessive financial burden.
Competitive Landscape and Key Companies
The anesthesia drugs market in the United States is competitive, with several major pharmaceutical and medical technology companies operating in the space. These companies focus on innovation, product development, and regulatory approvals to maintain market leadership.
Key players in the market include:
Baxter International Inc
AbbVie
Medtronic Plc
Draegerwerk AG
Koninklijke Philips NV
Abbott Laboratories
These organizations are involved in the development of anesthesia drugs, delivery systems, monitoring technologies, and related medical solutions that support surgical care.
Final Thoughts
The United States anesthesia drugs market is expected to maintain steady growth through 2033 as surgical procedures continue to rise and medical technologies advance. With increasing demand for safe, efficient, and patient-friendly anesthesia solutions, pharmaceutical companies are investing heavily in innovative drug formulations and improved delivery methods.
Government healthcare programs, strong regulatory oversight, and advancements in surgical technologies further support market expansion. As hospitals and surgical centers focus on improving patient outcomes and reducing recovery times, anesthesia drugs will remain an indispensable component of modern healthcare.
Looking ahead, the combination of medical innovation, growing surgical demand, and patient-centered healthcare practices will continue to drive the evolution of the anesthesia drugs market in the United States.






Comments
There are no comments for this story
Be the first to respond and start the conversation.